.
AG Gründker
|
|
|
GPER1
|
Der G-Protein gekoppelte Östrogenrezeptor 1 (GPER1)
in gynäkologischen Tumoren
Mammakarzinom:
Triple-negative
Mammakarzinome (TNBCs) exprimieren häufig den G-Protein-gekoppelten
Östrogenrezeptor 1 (GPER1), auch bekannt als G-Protein-gekoppelter
Rezeptor 30 (GPR30). GPER1 vermittelt nicht-genomische Wirkungen von
17β-Östradiol. Darüber hinaus binden der selektive
Östrogenrezeptormodulator (SERM) Tamoxifen und der komplette
ERα-Antagonist Fulvestrant an GPER1 und induzieren unerwünschte
Wirkungen in Brustkrebszellen. GPER1 wird teilweise für die
Entwicklung einer Resistenz gegenüber Tamoxifen verantwortlich gemacht.
Die Hemmung des GPER1 könnte eine aussichtsreiche Strategie zur
Therapie von TNBCs darstellen.
Zervixkarzinom:
GPER1 spielt eine wichtige Rolle bei Progression, Migration und
Therapieresistenz von Tumoren. Auf die Frage, ob GPER1
tumorunterstützend oder tumorunterdrückend wirkt, lassen sich in der
aktuellen Forschung vielfältige Antworten finden. Die Funktion von
GPER1 beim Zervixkarzinom (CC) ist bisher wenig verstanden.
GPER1 scheint beim CC eine tumorsuppressive Rolle zu spielen, denn die
Suppression von GPER1 führt zu einer verstärkten Klonogenität sowie zu
einem erhöhten invasiven Verhalten. Darüber hinaus kommt es zu einer
erhöhten Expression des Onkogens SERPINE1/PAI-1. Auch die
epithelial-mesenchymale Transition (EMT) scheint in Zellen mit
reduzierter GPER1-Expression verstärkt zu sein. GPER1 scheint demnach
im CC als Tumorsuppressor zu wirken. Dies deckt sich mit den
Ergebnissen einer klinischen Studie, die besagt, dass die Expression
von GPER1 mit einer positiven Prognose bei Patienten mit CC korreliert.
Eine andere Studie zeigt jedoch, dass eine hohe GPER1-Expression bei CC
mit einer schlechten Prognose verbunden ist. In dem einem aktuellen
Projekt untersuchen wir, inwieweit die GPER1 Expressionslevel seine
Funktion als Tumorsuppressor oder Onkogen bestimmen. In diesem
Zusammenhang spielt die komplexe Signaltransduktion des GPER1 (Abb. siehe unten)
eine wichtige Rolle. Es ist sehr wahrscheinlich, dass eine veränderte
Signaltransduktion aufgrund unterschiedlicher GPER1-Expressionsniveaus
dafür verantwortlich ist, ob GPER1 tumorsuppressiv oder tumorfördernd
wirkt.
Ovarial- und Endometriumkarzinom:
GPER1 ist an mehreren Signalwegen beteiligt (Abb. siehe unten),
die Tumorentwicklung und -progression regulieren. Aktuelle Daten deuten
darauf hin, dass GPER1 beim Ovarialkarzinom (OC) onkogen oder
tumorsuppressiv wirken kann, je nach dem spezifischen Zusammenspiel der
aktivierten und inaktivierten Signalwege in den Tumorzellen.
Obwohl die Rolle von GPER1 bei der Entwicklung von
Endometriumkarzinomen (EC) kontrovers diskutiert wird, deuten die
meisten Ergebnisse auf eine tumorfördernde Rolle im EC hin.
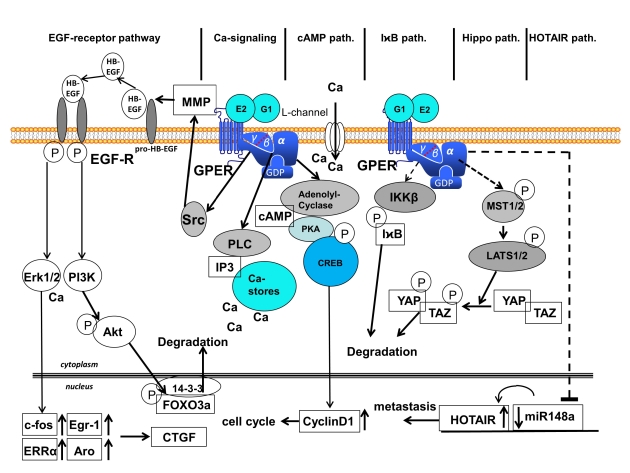
Signalwege, die nach Bindung von 17β-Östradiol (E2) oder dem Agonisten G1 am G-Protein-gekoppelten Östrogenrezeptor 1 (GPER1) aktiviert werden.
Es werden sechs verschiedene Signalwege unterschieden: (1)
EGF-Rezeptor, (2) Kalzium, (3) cAMP, (4) IχB, (5)
Hippo und (6) HOTAIR Signalweg.
Girgert R, Emons, G, Gründker C. Frontiers in Endocrinology 2019;9:781
. Publikationen:
- Schmitz
V, Bauerschmitz G, Gallwas J, Gründker C (2022) Suppression of G
protein-coupled estrogen receptor 1 (GPER1) enhances the anti-invasive
efficacy of selective ERβ agonists. Anticancer Research 42(11):5187-5194
- Girgert R, Emons, G, Gründker C (2019) Estrogen signaling in ERα-negative breast cancer: ERβ and GPER. Frontiers in Endocrinology 9:781
-
Girgert
R, Emons G, Gründker C (2018) Inhibition of growth hormone
receptor by Somavert reduces expression of GPER and prevents growth
stimulation of triple-negative breast cancer by 17β-estradiol. Onology Letters 15(6):9559-9566
-
Girgert R, Emons G, Gründker C (2017) Gefitinib inhibits
growth of triple-negative breast cancer cells in part by reduction of GPER
expression, preventing 17β-estradiol
induced cell growth. Oncology Reports 37(2):1212-1218 -
Hinsche O, Girgert R, Emons G, Gründker C (2015) Estrogen receptor β selective agonists reduce invasiveness of triple-negative breast cancer cells. International Journal of Oncology 46:878-884
-
Girgert R, Emons G, Gründker C (2014) Inhibition of
GPR30 by estriol successfully prevents growth stimulation of triple-negative
breast cancer cells by 17β-estradiol. BMC Cancer 14:935
-
Girgert R, Emons G, Gründker C (2012) Inactivation
of GPR30 reduces growth of triple-negative breast cancer cells: possible
application in targeted therapy. Breast Cancer Research and Treatment 134:199-205
- Block M, Gründker C, Fister S, Kubin J,
Wilkens L, Mueller MD, Hemmerlein B, Emons G, Günthert AR (2012)
Inhibition of the AKT/mTOR and erbB pathways by gefitinib, perifosine
and analogs of gonadotropin-releasing hormone I and II to overcome
tamoxifen resistance in breast cancer cells. International Journal of Oncology 41:1845-1854
-
Block M, Fister S, Emons
G, Seeber S, Gründker C, Günthert AR (2010) Antiproliferative effects of antiestrogens
and inhibitors of growth factor receptor signaling on endometrial cancer. Anticancer
Research 30(6):2025-2031
-
Günthert AR, Gründker
C, Olota A, Läsche J, Eicke N, Emons G (2005) Analogs of gonadotropin-releasing hormone
I (GnRH-I) and GnRH-II inhibit EGF-induced signal transduction and resensitize resistant
human breast cancer cells to 4OH-tamoxifen. European Journal of Endocrinology 153:613-625
|
|
|
|
|

